News
Explore News
Latest News
-

PS Plus adding stunning action-RPG for free I would've paid good money for
Patience is a virtue
By Rik Henderson Published
-

Your Apple TV just got a great new trick
Actually it got a couple...
By Britta O'Boyle Published
-

Denon's keeping CDs alive with a new high-end player
While other firms desert discs, Denon continues to show faith in those shiny circles
By Carrie Marshall Published
-

Lockin’s new smart lock doubles as a video doorbell and reads your palms
Lockin launches the 4-in-1 Smart Lock and its recognition is seriously impressive
By Bethan Girdler-Maslen Published
-

Your O2 Android phone will now come with Epic Games Store preinstalled
Gaming on your phone just got easier
By Sam Cross Published
-

Google Pixel 9a exposed – major leak reveals specs, price and more
Google’s affordable Pixel phone for 2025 shares all
By Chris Hall Published
-
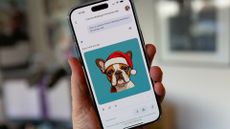
Gemini 2.0 announced – one step closer to being the ultimate assistant
Android phones are about to get a powerful update
By Sam Cross Published
-

Apple just accidentally leaked the M4 MacBook Air
Don't expect a redesign, this upgrade is all about processing power
By Carrie Marshall Published
-

Xiaomi's new mini washer dryer has 20 different programmes – and it's only 18 inches tall
It's the ideal appliance for smaller living spaces
By Lizzie Wilmot Published
